Home > Capabilities > Manufacturing > Insulin & Insulin Analogs
Insulin & Insulin Analogs
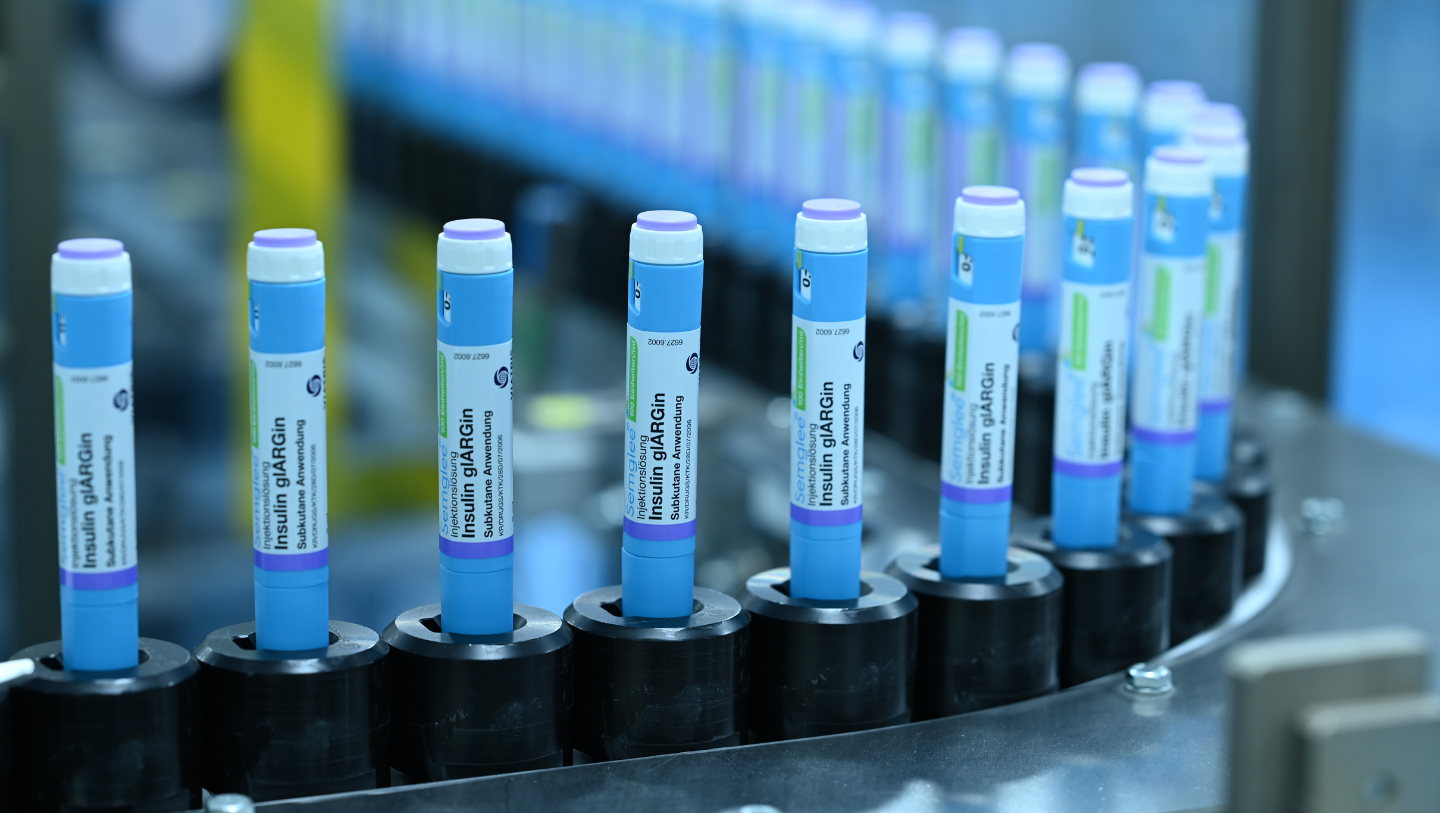
Biocon Biologics has established itself as a leader in insulin and insulin analog manufacturing, leveraging cutting-edge technology and global-scale infrastructure across our facilities in Bengaluru, India, and Johor, Malaysia

The proprietary Pichia pastoris platform at the core of our insulin production, enables the large-scale expression of recombinant human insulin (rh-Insulin) and insulin analogs. This innovative approach led to the successful launch of Biocon’s first insulin brand, Insugen®, in 2004, marking a pivotal moment in the company’s entry into the branded formulations market. Our focus on innovation is exemplified by advanced fermentation and multi-stage purification processes that ensure the production of high-purity human insulin and analog drug substances, which are converted into drug product variants (soluble and suspensions) and also assembled into pens.
Asia’s Largest Integrated Insulin Manufacturing Plant
Biocon SDN. BHD., Malaysia, was established as Biocon Biologics’ first overseas manufacturing facility. Located within BioXcell, a biotechnology park in Iskandar Puteri, Johor, the facility has end-to-end capabilities for manufacturing a robust portfolio of human insulin and insulin analogs for global markets.

Our state-of-the-art insulin manufacturing center at Johor, Malaysia, is recognized as Asia’s largest integrated insulin manufacturing plant.
Related Links
Biocon Biologics is a leader in the science, scale, and complexity of manufacturing monoclonal antibodies (mAbs) and conjugated proteins. Over...