
Home > Capabilities > Research & Development > Clinical Development
Clinical Development
Our Global Medical Affairs team connects Biocon Biologics with the healthcare ecosystem by delivering accurate, evidence-based scientific data on our biosimilars and biologics.
By engaging healthcare professionals, regulatory bodies, and Key Opinion Leaders (KOLs), the team advances clinical understanding, supports the development of guidelines, and promotes the adoption of biosimilars for equitable, sustainable healthcare.
From leading medical education programs to hosting global webinars and scientific conferences, we foster knowledge exchange that builds trust and drives better patient outcomes.
Clinical Development
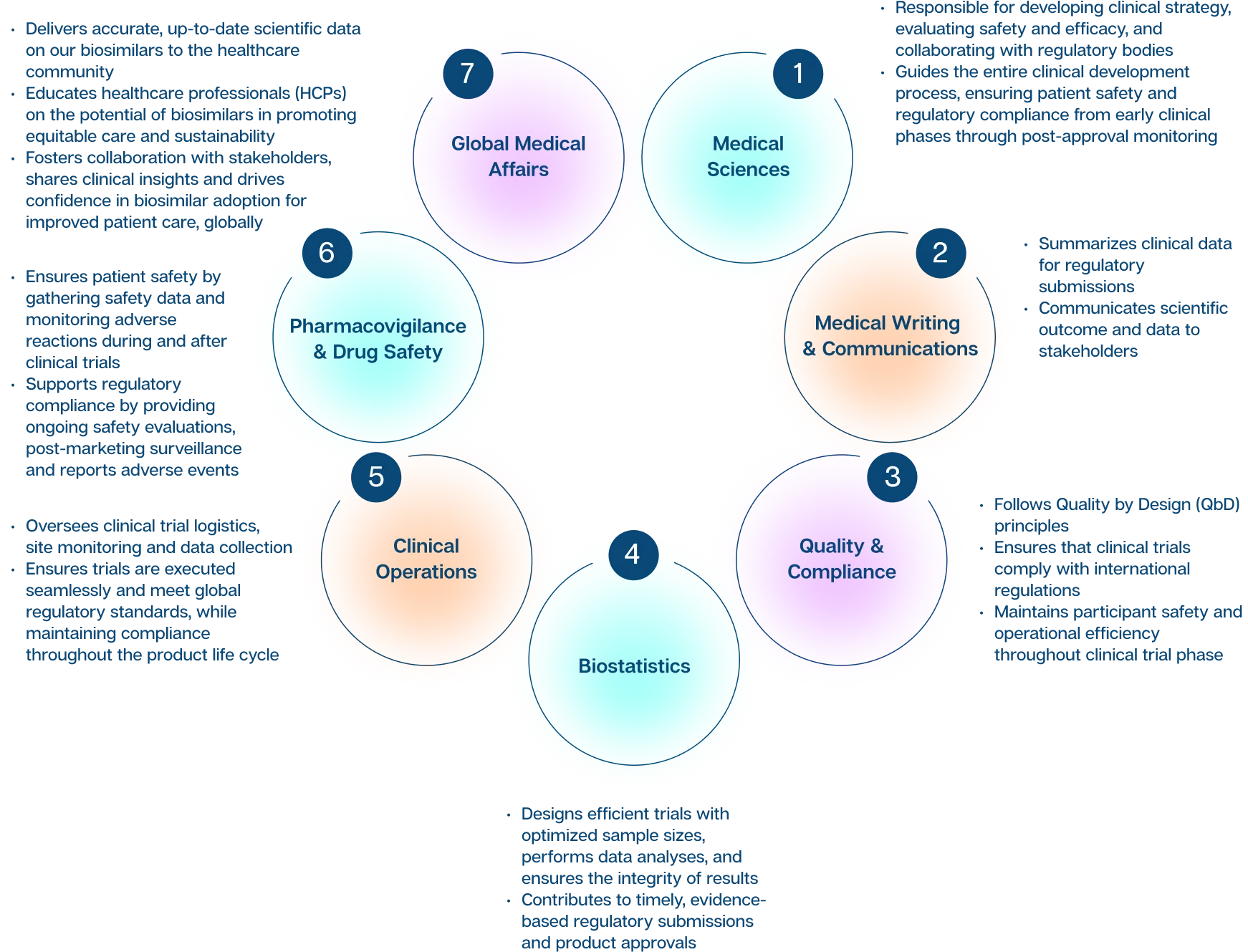
Global Medical Affairs
The Global Medical Affairs team bridges the gap between Biocon Biologics and the healthcare community by delivering accurate, up-to-date scientific data on our biosimilars.
The team is responsible for educating healthcare professionals (HCPs) on the potential of biosimilars to promote equitable care and sustainability, playing a pivotal role in advancing the scientific and clinical understanding of our biosimilars and biologics portfolio. In addition to supporting the development of clinical guidelines in close collaboration with regulatory bodies, it leads key medical education initiatives, including webinars and scientific conferences, to foster knowledge exchange and drive innovation.
Through active engagement with Key Opinion Leaders (KOLs) and stakeholders, Global Medical Affairs facilitates collaboration, shares clinical insights, and drives confidence in the adoption of biosimilars, for improved patient care.

