
Home > Capabilities > Research & Development > Regulatory Sciences
Regulatory Sciences
Biocon Biologics’ Regulatory Sciences team drives global biosimilar approvals by mastering and shaping the evolving regulatory landscape. We design globally aligned regulatory strategies—spanning abbreviated pathways, comparability, interchangeability, and indication extrapolation—for insulins, monoclonal antibodies, and complex protein biologics.
Located at our Bengaluru and Chennai R&D hubs, our regulatory experts coordinate worldwide filings—from IND/CTA to BLA/MAA—in ICH-regulated and emerging markets. With successful dossier submissions across the US FDA, EMA, TGA, Canada, Brazil, Japan, and more, we are recognized as a trusted voice in biosimilars, accelerating approvals that support affordable patient access
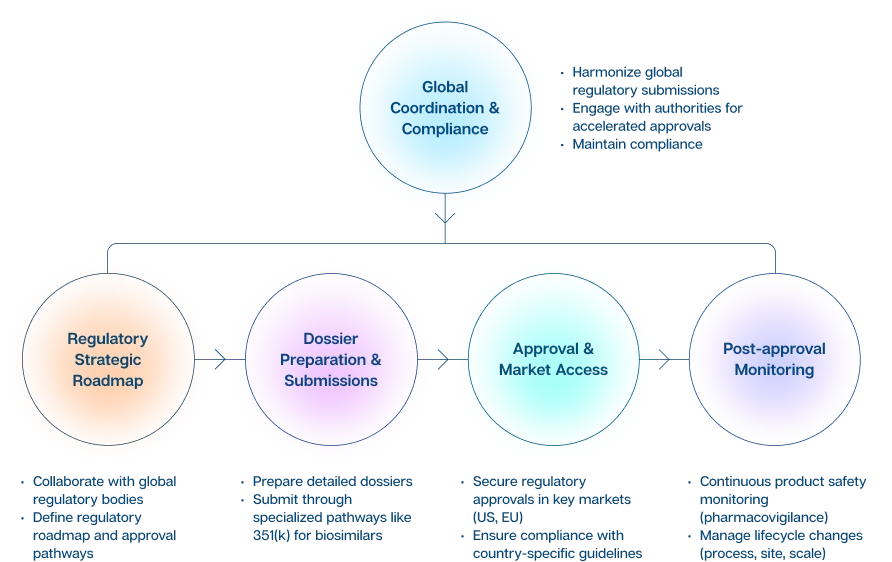
Regulatory Affairs: Key Functions
Biocon Biologics has achieved many global ‘firsts’ and set new benchmarks for the global biosimilars industry.
First Indian company to introduce a biosimilar (bGlargine) in Japan’s highly regulated market, in 2016.
First company globally to receive U.S. FDA approval for biosimilar Trastuzumab in 2017 and biosimilar Pegfilgrastim in 2018.
First company globally to commercialize interchangeable bGlargine in the U.S.
First company to get U.S. FDA approval for interchangeable bAflibercept.

